Research Highlights

Great Lakes Bioenergy researchers and collaborators engineered softwoods to incorporate a key feature of hardwoods. The resulting pine (shown here) processes more easily into pulp and paper.
Great Lakes Bioenergy research consistently results in new discoveries and new technologies. Here, we highlight high-impact research from all three of our research areas.
Drought timing affects switchgrass metabolism, biofuel yields
Switchgrass is a promising bioenergy crop, due in part to its resilience to drought stress, but there is limited information on how drought affects metabolite profiles across developmental stages, in particular the distinction between core and developmentally specific physiological and metabolic drought responses. So researchers subjected plants to four watering treatments at specific developmental stages and measured the impact on growth and downstream ethanol yields.
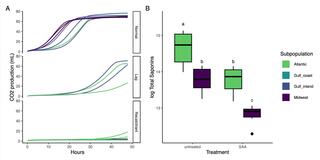
Switchgrass steroidal saponins that reduce fungal disease in the field inhibit yeast fermentation
Scientists measured variation in field fungal infection, specialized metabolite production, and fermentation efficiency in 102 genotypes of switchgrass and investigated methods for rescuing fermentation for poorly fermenting lines.
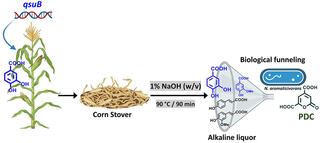
Engineered accumulation of protocatechuate in corn biomass can enhance biomanufacturing
Protocatechuate (DHBA) serves as a precursor for a wide range of valuable bioproducts. Crops can accumulate DHBA via expression of dehydroshikimate dehydratase (QsuB). This experiment sought to overproduce DHBA in plant biomass for downstream biological upgrading.
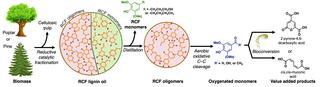
Cu-catalyzed oxidation cleaves C–C bonds in lignin-derived oligomers for biological funneling
Most methods for lignin deconstruction cleave carbon–oxygen bonds, resulting in suboptimal yields of aromatic monomers and formation of oligomers with intact carbon–carbon bonds that limit the yield of valorized products. This work demonstrates a copper-catalyzed aerobic oxidation process that converts oligomeric “waste” derived from reductive catalytic fractionation (RCF) of woody biomass into aromatic monomers. The resulting monomers are used as feedstock for biological funneling into bioproducts.
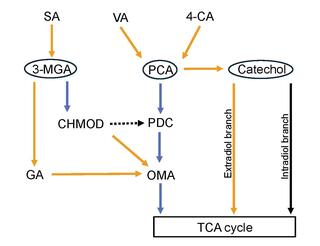
Systems-level analysis can improve aromatic funneling by N. aromaticivorans and other bacteria
Researchers performed a systems-level analysis of a LysR-family transcription factor in Novosphingobium aromaticivorans, which could inform engineering of commercially relevant bacteria that can remove toxic aromatics or metabolize them into industrial chemicals.
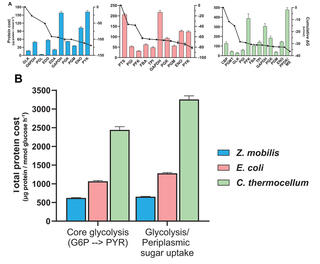
Living evidence reveals how thermodynamics shape enzyme investment in glycolysis
Computational studies predict that thermodynamically constrained reactions and pathways impose greater protein demands on cells, requiring a larger amount of enzyme to sustain a given flux compared to those with stronger thermodynamics. This study provides in vivo evidence that thermodynamic driving forces are a key parameter influencing the enzyme burden of metabolic pathways.
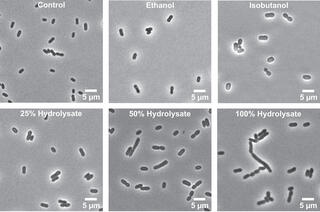
Lignocellulosic hydrolysate exposure elicits unique metabolic shifts in Zymomonas mobilis
Despite growing interest in Zymomonas mobilis as a biocatalyst, its physiological response to lignocellulosic hydrolysates remains poorly understood. Here researchers investigated the physiological response of Z. mobilis to ammonia fiber expansion (AFEX)-pretreated switchgrass hydrolysate using a systems-level approach integrating LC–MS/MS-based lipidomics and shotgun proteomics.
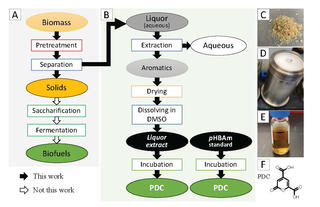
Bioconversion of phenolics in biomass spent liquor into industrial chemicals
Spent liquors from biomass pretreatment could provide a source for renewable chemical production. This study characterized the phenolics in spent liquor from aqueous ammonia pretreatment of poplar, investigated the impact of metal additives on the formation of phenolics during pretreatment, and explored valorization microbial transformation.
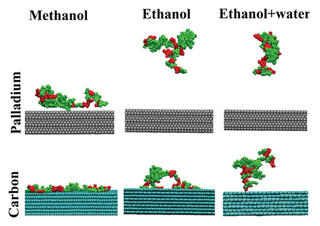
Molecular dynamics simulations reveal effects of solvent on lignin-surface interactions
Scientists performed all-atom molecular dynamics simulations to study lignin solvation, solvent-mediated conformational changes, and the interaction of solvated lignin oligomers with model surfaces. They focused on the behavior of a model lignin oligomer in methanol, ethanol, a binary mixture of ethanol and water, and water alone at typical RCF reaction temperature (473 K) and room temperature. Model palladium (Pd) and carbon (C) surfaces were introduced to understand how solvent choice impacts adsorption onto a representative catalytic surface and to quantify competition among reactant and solvent molecules for surface binding sites.
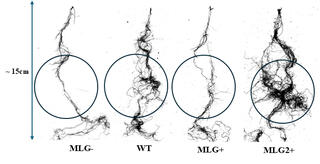
Increased MLG in engineered grasses changes roots, may affect soil carbon processing
Researchers are pursuing efforts to design grasses with elevated levels of cell wall carbohydrates, including mixed-linkage glucan (MLG), to improve their value as bioenergy feedstocks. By studying four grasses with varying MLG production, scientists showed that strains engineered for higher MLG production also accumulate MLG in the roots, potentially affecting soil carbon cycling.