Triple-transgenic trees show altered lignin composition and better deconstruction
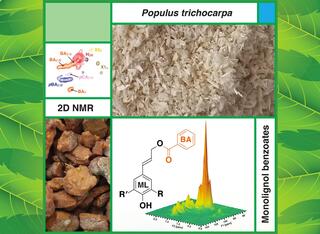
Downregulation of three key monolignol biosynthetic genes added a new ‘clip-off’ to lignins, reduced total lignin content, and increased sugar release.
The Science
Lignin, an abundant renewable aromatic biopolymer in terrestrial vascular plants, is typically synthesized from three monolignols: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. However, the plasticity of the lignification process allows plants to shift the relative abundance of different subunits or add new structures. In this collaborative paper, Great Lakes Bioenergy Research Center (GLBRC) scientists and colleagues at North Carolina State University produced triple-transgenic poplar with reduced expression of three lignin biosynthetic cytochrome P450 genes. Lignin from these transgenics had significantly higher levels of monolignol benzoates (ML-BA) as well as additional desirable traits, including reduced lignin content and improved saccharification efficiency. The results suggest that benzoate biosynthesis may be a useful target to improve the value of lignin ‘clip-offs’ as well as the production of sugars from biomass.
The Impact
Identifying and characterizing novel lignin structures are crucial to understand the range of potential lignin precursors and to elucidate the effects of such variation on the physical and chemical properties of lignin biopolymers. This work represents a valuable advance in understanding the complexity and capability of lignin biosynthetic pathways. The transgenic poplar produced in this study exhibits improved traits, including: 1) higher levels of new simple phenolic esters that can be easily clipped off from the transgenic lignin to add value to a biorefinery operation; and 2) lower total lignin content, resulting in improved sugar release.
Summary
Genetic modifications in the lignin biosynthesis pathway are a promising avenue for improving lignin and biomass quality and for producing high-value commodity chemical feedstocks. Benzoate conjugates are important metabolites in plants that have not been adequately characterized as components of the lignin structure. This study found that these conjugates occur at trace levels in wild-type poplar lignin and were significantly augmented in triple transgenics with downregulated genes for three key cytochrome P450 enzymes, two 4-hydroxylases (PtrC4H1 and PtrC4H2), and one 3-hydroxylase (PtrC3H3). Monolignol benzoate conjugates were incorporated into the transgenic lignin at a level some 16-fold higher than in wild-type. The co-downregulation of the three P450 genes may cause a rerouting or modification of the pathway due to the interaction of the benzoate biosynthetic pathway with the conventional pathway for monolignol biosynthesis. The total lignin content in the transgenics was decreased by 50%, and the lignin H-unit level was elevated about 70-fold. The wood from the transgenic trees showed reduced recalcitrance toward enzymatic saccharification, which has value for the conversion of plant biomass to bioenergy. The findings indicate that the benzoate biosynthetic pathway should be considered a legitimate segment of the monolignol biosynthetic pathway in poplar.
Contacts (BER PM)
N. Kent Peters
kent.peters@science.doe.gov, 301-903-5549
Corresponding Authors
Hoon Kim
University of Wisconsin–Madison
hoonkim@wisc.edu
John Ralph
jralph@wisc.edu
Vincent L. Chiang
North Carolina State University
vchiang@ncsu.edu
Funding
This work was supported by grants from the DOE Great Lakes Bioenergy Research Center (DOE BER Office of Science DE-SC0018409) and the National Science Foundation Plant Genome Research Program grant (DBI-0922391).
Publication
H. Kim et al. “Monolignol benzoates incorporate into the lignin of transgenic Populus trichocarpa depleted in C3H and C4H.” ACS Sustainable Chemistry & Engineering (2020) 8, 3644-3654 [DOI: 10.1021/acssuschemeng.9b06389]
Related Link
https://pubs.acs.org/doi/abs/10.1021/acssuschemeng.9b06389