Researchers engineer plant oils to make—and store—valuable organic compounds

Bioenergy crops are a potential source of renewable alternatives for many petroleum-derived products. Great Lakes Bioenergy Research Center (GLBRC) scientists at Michigan State University (MSU) have now devised a system to coax plants into producing higher amounts of two valuable organic materials, terpenes and lipid droplets, and package them together inside cells for easy extraction.
What’s that smell?
There’s nothing like the experience of hiking through coniferous woods – the soft crunch of needles underfoot, the silhouette of the forest canopy against the sky, and of course, the sweet smell of terpenes in the air.
Among their many functions, terpenes deliver the pungence associated with evergreens. Plants naturally make these oily substances to fend off invaders, attract friendly pollinators, and even rehabilitate injury. Terpene derivatives, called terpenoids, are versatile as commodity chemicals for manufacturing, as well as fragrances and oils.
Synthetic versions of these compounds can be made out of crude oil, but the cost of doing so is prohibitive for large-scale production. The ability to extract higher quantities of terpenes from natural plant sources would increase access to these useful industrial precursors while simultaneously giving an economic boon to the biofuels industry.
Changing pathways
Bjoern Hamberger, MSU assistant professor of biochemistry and molecular biology, is part of a GLBRC research group focused on increasing terpene production in plants.
In a new study, published today (February 20, 2019) in the journal Nature Communications, Hamberger and an MSU team outline a series of steps to boost plants’ ability to produce several useful terpenoids, including abietic acid, found in the sticky sappy substance that firs emit when they make that familiar woodland smell.
Radin Sadre, synthetic biologist and biochemist in MSU's department of horticulture, is the lead author of the study devised by Hamberger and Christoph Benning, MSU-DOE Plant Research Laboratory director. Benning’s previous GLBRC work on increasing lipid production in plants was the genesis of the current research.
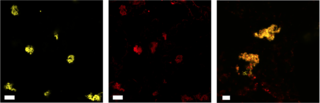
The GLBRC scientists at MSU embedded the tobacco relative Nicotiana benthamiana, a species with a well-understood genetic makeup, with genes from known terpene superproducers via a soil bacterium that naturally inserts DNA into the plant. They also selected enzymes known for their stability, including one from a kind of bacteria that only survives in temperatures hotter than 55 degrees Celsius.
The result was an N. benthamiana plant that manufactured much higher levels of several kinds of terpenoids—in one case, up to a 360-fold increase—anchored to lipid droplets for easy extraction.
Like mixing oil with oil
“When you have cells that are rampant producers of terpenoids, like we’ve produced, at some point these molecules will start disrupting other cellular processes,” says Hamberger.
To hold all those extra compounds in N. benthamiana, the GLBRC team bumped up production of lipid droplets, structures that normally help store and regulate fatty molecules in the cell. The surplus lipid droplets helped trap the terpenes, making them easier to cull from the plant.
“Lipid droplets are an oily environment. The terpenoids we are overproducing don’t mix with water, so the lipid droplets were a perfect spot to anchor them,” Hamberger explains.
As a bonus, lipid droplets provide a second valuable bioproduct because they can be converted into high-octane fuels.
N. benthamiana is akin in biological makeup to poplar, a fast-growing tree that GLBRC researchers have targeted as a bioenergy crop for growth on lands not suitable for traditional agriculture. Working with a similar cellular machinery paves the way for Hamberger to try some of the best terpene-boosting genes in poplar without the same hassle as starting from a less-similar organism.
“Our big vision is to engineer these biomass species with new traits which will increase their worth,” says Hamberger. “Lipid droplets are perfect solvents for terpenoids – when we extract the former, we’re now getting the latter for free.”
MSU researchers contributing to this study include Peiyen Kuo, Jiaxing Chen, Yang Yang and Aparjita Banerjee. This work was primarily supported by the U.S. Department of Energy-Great Lakes Bioenergy Research Center Cooperative Agreement DE-FC02-07ER64494 (B.H., C.B.) and partially by the Division of Chemical Sciences, Geosciences and Biosciences, Office of Basic Energy Sciences of the U.S. Department of Energy Grant DE- DE-FG02-91ER20021 (C.B.) and by AgBioResearch MICL02357 (C.B.), Michigan State University. B.H. gratefully acknowledges startup funding from the Department of Molecular Biology and Biochemistry, AgBioResearch (MICL02454) and the U.S. Department of Energy Grant (DE-SC0018409), Michigan State University.