Novel cellulose form requires fewer enzymes for better biomass breakdown
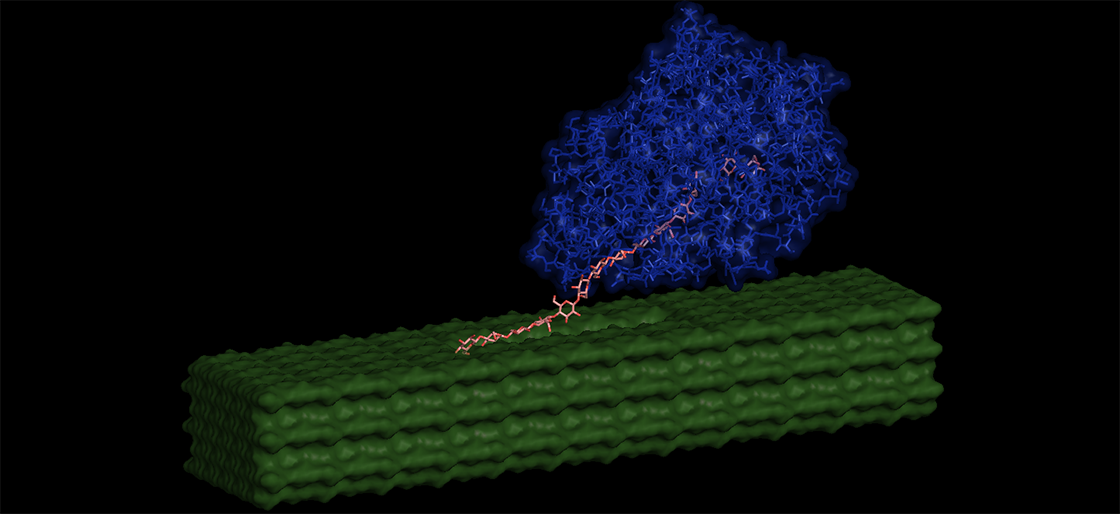
A new biomass breakdown method being developed by researchers at the Great Lakes Bioenergy Research Center (GLBRC) and the Los Alamos National Laboratory (LANL) has the potential to improve biofuel production by making it significantly easier for enzymes to release sugars from plant tissues.
In a June Proceedings of the National Academy of Sciences article, researchers demonstrated that a novel biomass pretreatment using ammonia can modify cellulose—a key plant structural component—allowing up to five times more sugar to be released from the biomass by enzymes.
Surprisingly, this improvement occurred even though fewer enzymes were able to latch onto the surface of the modified cellulose.
GLBRC’s Shishir Chundawat likens the crystalline cellulose nanofibers in plant cell walls to steel rod-like reinforcements embedded in concrete. While the nanofibers give the cell wall rigidity, they ultimately limit enzyme access to cellulose.
“One of the keys to cheaper biofuel production is to unravel these nanofibers more efficiently into soluble sugars using fewer enzymes,” says Chundawat.
From a previous study, Chundawat and his colleagues already knew that a process called ammonia pretreatment produces a modified form of cellulose, dubbed “cellulose III,” that greatly facilitates the process of sugar extraction. However, the fact that this occurred even with reduced enzyme binding seemed counterintuitive.
It was almost as though deconstructing unmodified cellulose was equivalent to working a steady job, while deconstructing cellulose III was like winning the lottery: not very many enzymes succeeded in binding to cellulose III’s surface, but those that did got a much bigger payoff in the form of increased sugar production.
“Forming cellulose III changes the physical characteristics of the surface of cellulose molecules, and native enzymes have not evolved to deal with it,” says Chundawat, explaining that the surface of the modified cellulose is “less sticky” than that of regular cellulose, and therefore harder for the deconstruction enzymes to grab onto.
Fortunately, it seemed that the same physical changes that made it hard for enzymes to bind to cellulose III also helped them unravel its nanofibers with ease. To explore this idea, GLBRC researchers collaborated with LANL modeling experts to simulate what happens when an enzyme from the fungus Trichoderma reesei binds to and hydrolyzes crystalline cellulose.
The researchers theorized that under certain conditions, the enzymes could indeed untangle simple carbohydrate molecules from cellulose III much faster than they could from native cellulose despite reduced binding. The model that the researchers used also helped them to better understand the interactions between enzymes and cellulose III that affected how cellulose molecules were broken down into soluble sugars.
The researchers estimate that using ammonia pretreatment to convert cellulose from bioenergy feedstocks to cellulose III could significantly reduce the amount of expensive enzyme needed for biofuel processing; however, they are already looking for ways to improve the process.
Chundawat, who until recently worked with GLBRC’s Bruce Dale at Michigan State University where the ammonia pretreatment technology is being developed, now works with Deconstruction Area Co-Leader Brian Fox in the University of Wisconsin-Madison Department of Biochemistry. Fox and Chundawat aim to better understand the dynamics of cellulose III-enzyme interactions, and to develop novel enzymes that are even more effective at breaking down cellulose III.
“In the long term, we are hoping to further increase the rate of [biomass deconstruction] by engineering more effective pretreatments and enzymes,“ Chundawat says. Achieving rapid deconstruction while reducing enzyme requirements could make for a highly efficient, low-cost biofuel production method—an energy researcher's jackpot.