Glowing green: Colorful new method reveals secrets of plant metabolism
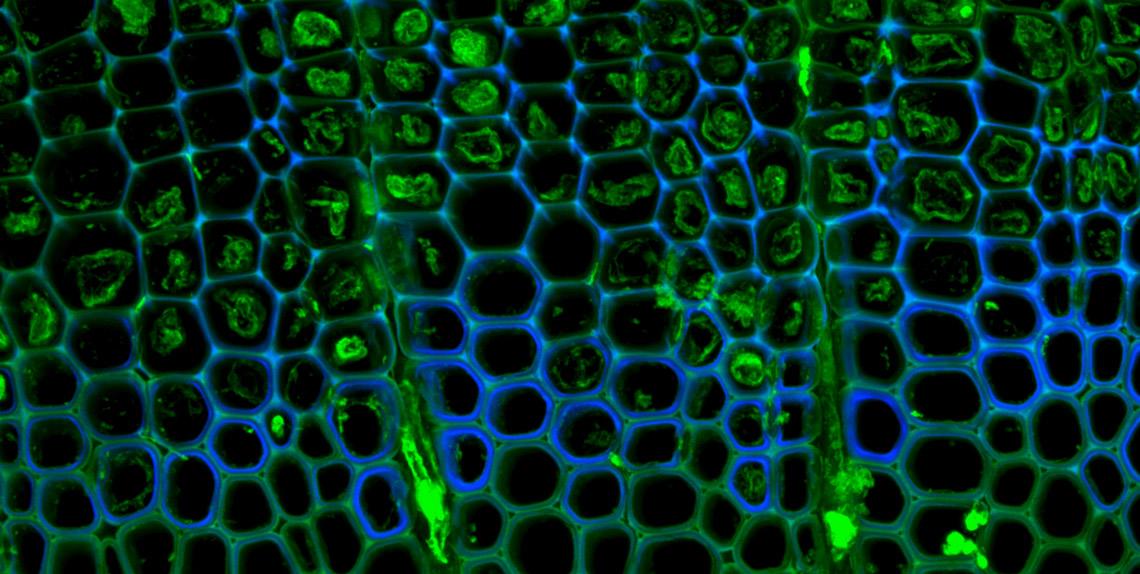
Trying to understand how the structural polymer lignin is deposited in plant tissues is a bit like trying to understand bone growth in a human: it is very tricky to observe in a living specimen. But Yuki Tobimatsu of the University of Wisconsin-Madison has found a way: by tagging molecular building blocks with brightly colored dyes which, when exposed to ultraviolet light, fluoresce brilliantly.
The new method, which was recently published in The Plant Journal, offers a rare in vivo glimpse of plant metabolism, and could have far-reaching applications for many areas of science in addition to bioenergy.
"A big question in plant biosynthesis is how molecules are transported and combined into polymers," says Tobimatsu, a researcher in the Great Lakes Bioenergy Research Center (GLBRC) plants research area. "Our tagged molecules can be used to discover such mechanisms, because the dye markers can be taken up by plant cells and incorporated into their metabolic pathways."
Tobimatsu explains that the molecules, called monolignols, are lignin precursors. Tagging monolignols with dye allows researchers to follow them through the tissues of living plant seedlings as they are absorbed and utilized.
"The cool thing about Yuki's dye is that it only stains plant tissues that are metabolically active, so only the parts of the plant that are actually producing lignin change color," says John Ralph, a UW-Madison professor of biochemistry and biological systems engineering and leader of GLBRC Plants research.
Because lignin is a structural barrier to processing cellulosic biomass into ethanol, biofuel researchers are always interested in learning more about its properties to improve technologies for breaking it down. However, Ralph and Tobimatsu say that their new tool has many potential applications for plant scientists, and they have already received several requests for their tagged monolignols from external research groups.
Researchers could potentially tag other molecules with fluorescent red, green, or blue dyes to make plant tissues stand out when photographed, without disrupting their normal functioning.
"The best thing about this process is that Yuki attaches the dyes to the monolignols in a way that doesn't interfere with their biological performance...they just do what they normally do," says Ralph.
The research was conducted in collaboration with several research institutions around the world, including the DOE Joint BioEnergy Research Center (JBEI), the Scion research institute in New Zealand, Ghent University in Belgium, and Purdue University.