GLBRC and JBEI work together to break down lignin, advance biofuels
To tackle what many consider the next frontier in biofuels research, the Great Lakes Bioenergy Research Center (GLBRC) recently joined forces with the Joint BioEnergy Institute (JBEI) in Emeryville, California. The focus of their collaboration? Lignin, a glue-like compound in the cell wall of most living plants that gives them their sturdiness.
While lignin accounts for up to one third of plant carbon, it's also the most difficult part to break down and remains a formidable obstacle to accessing the valuable sugars contained within biomass. As a result, the biofuels and paper industries mostly treat it as a waste product, isolating it to be burned or discarded.
But GLBRC and JBEI researchers hope to change all that by bringing their collective expertise to the challenge of unraveling lignin’s strong and unique molecular bonds. With two new studies already complete, the payoff for their effort could be significant.
“If we can convert lignin from an undesirable byproduct into a starting material for advanced biofuels and other lucrative chemicals,” says Tim Donohue, GLBRC director and UW–Madison professor of bacteriology, “we would dramatically change the economics of tomorrow’s biorefineries.”
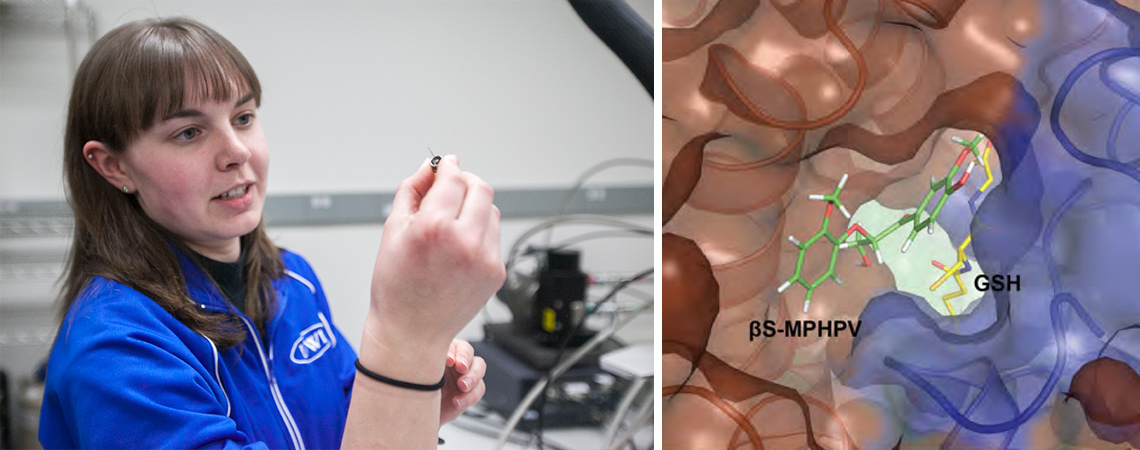
The researchers began by studying bacterial enzymes that cleave specific chemical bonds inside larger lignin molecules with the idea of creating a single new enzyme to break down lignin. That turned out to be a difficult task, however, because lignin molecules are similar to snowflakes: they have a multitude of spatial configurations, even within a single plant, and each of them requires a different approach to deconstruction.
“Making a single enzyme would be like trying to make a glove that’s designed for your left hand fit on your right hand,” explains Kate Helmich, co-first author of one of the studies and a recent PhD graduate of UW–Madison’s biochemistry department. “Our two hands are different configurations of the same fingers, and lignin is like a chain of many different hands. Degrading that entire chain would require an enzyme, or glove, that can attach to both the left and the right hands within it.”
Nature has instead evolved multiple, highly specialized bacterial enzymes that work in concert to break down lignin, each of them cleaving different sets of chemical bonds in the complex molecule.
Though a single lignin-cracking “master hybrid enzyme” remains elusive, the study informs future enzyme engineering efforts and sets the stage for more GLBRC and JBEI collaborations to convert lignin and other parts of plant biomass into biofuels and valuable chemicals.
GLBRC’s Helmich and Daniel Gall share first authorship, with JBEI’s Jose Henrique Pereira, on a paper recently published in the Journal of Biological Chemistry; and former GLBRC researcher and Rice University professor George Phillips, Jr. is senior author. In a sister study led by JBEI (currently slated for publication), the team reports new insights into bacterial enzymes that could someday help produce aromatic compounds of great interest to the chemical industry from lignin.
“It is exciting to see that our collaboration has resulted in the simultaneous publication of these two important papers,” Donohue says. “Collaboration is at the center of everything we do at GLBRC. We look forward to doing more of this mutually beneficial work with JBEI and other academic and industrial partners to advance the growing biofuels industry.”
GLBRC is one of three Department of Energy-funded Bioenergy Research Centers created to make transformational breakthroughs that will form the foundation of new cellulosic biofuels technology. For more information on the GLBRC, visit www.glbrc.org.