pHBMT1, a BAHD-family monolignol acyltransferase, mediates lignin acylation in poplar
de Vries et al. "pHBMT1, a BAHD-family monolignol acyltransferase, mediates lignin acylation in poplar" Plant Physiology (2021) [DOI:10.1093/plphys/kiab546]
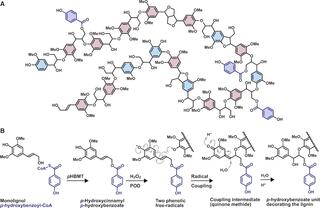
Poplar (Populus) lignin is naturally acylated with p-hydroxybenzoate ester moieties. However, the enzyme(s) involved in the biosynthesis of the monolignol–p-hydroxybenzoates have remained largely unknown. Here, we performed an in vitro screen of the Populus trichocarpa BAHD acyltransferase superfamily (116 genes) using a wheatgerm cell-free translation system and found five enzymes capable of producing monolignol–p-hydroxybenzoates. We then compared the transcript abundance of the five corresponding genes with p-hydroxybenzoate concentrations using naturally occurring unrelated genotypes of P. trichocarpa and revealed a positive correlation between the expression of p-hydroxybenzoyl-CoA monolig-nol transferase (pHBMT1, Potri.001G448000) and p-hydroxybenzoate levels. To test whether pHBMT1 is responsible for the biosynthesis of monolignol–p-hydroxybenzoates, we overexpressed pHBMT1 in hybrid poplar (Populus alba × P. grandidentata) (35S::pHBMT1 and C4H::pHBMT1). Using three complementary analytical methods, we showed that there was an increase in soluble monolignol–p-hydroxybenzoates and cell-wall-bound monolignol–p-hydroxybenzoates in the poplar transgenics. As these pendent groups are ester-linked, saponification releases p-hydroxybenzoate, a precursor to parabens that are used in pharmaceuticals and cosmetics. This identified gene could therefore be used to engineer lignocellulosic biomass with increased value for emerging biorefinery strategies.
Gene reports are available on Phytozome under accession numbers pHBMT1: Potri.001G448000. Putative pHBMTs: Potri.001G447300, Potri.001G447400, Potri.001G447500, Potri.011G153500.