Efficient synthesis of pinoresinol, an important lignin dimeric model compound
F. Yue et al. "Efficient synthesis of pinoresinol, an important lignin dimeric model compound" Frontiers in Energy Research 9 (2021) [DOI: 10.3389/fenrg.2021.640337]
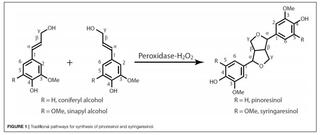
Pinoresinol is a high-value monolignol-derived lignan used in plant defense and with human health-supporting effects. The synthetic yield and isolation efficiency of racemic pinoresinol from coniferyl alcohol by conventional radical coupling methods is sub-optimal. In this work, a facile and efficient synthetic approach was developed to synthesize pinoresinol with much higher yield. By using 5-bromoconiferyl alcohol, which was synthesized in high yield from 5-bromovanillin, to make 5,5′-bromopinoresinol via a peroxidase mediated radical coupling reaction takes advantage of the smaller variety of radical coupling products from the 5-substituted monolignol, producing simpler product mixtures from which 5,5′-bromopinoresinol may be readily crystalized with good yield (total yield of 44.1% by NMR; isolated crystalline yield of 24.6%). Hydro-debromination of the crystalline 5,5′-bromopinoresinol to pinoresinol was essentially quantitative. Gram quantities of pinoresinol were conveniently synthesized by using this approach. This simple alternative pathway to make pinoresinol will impact pinoresinol-related research including structural characterization and modification of lignins, as well as clinical applications of pinoresinol and its derivatives.
The data presented in the study are included in the Supplementary Material.